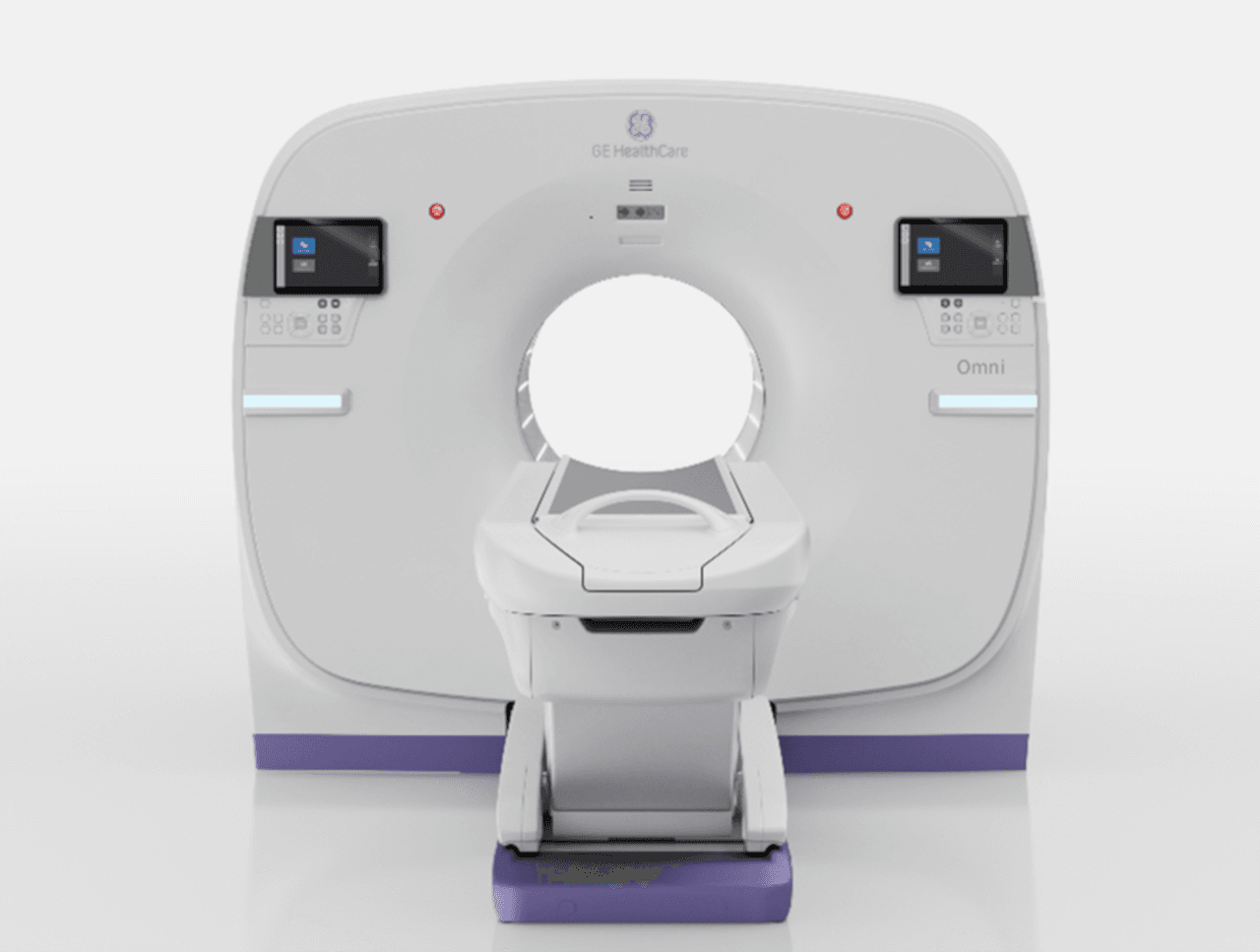
GE HealthCare installs Allia Moveo AI-assisted precision C-arm in France
Allia Moveo has received US Food and Drug Administration (FDA) 510(k) clearance and CE marking, bringing advanced mobility and precision to GE HealthCare’s interventional suite. The latest Allia platform assists clinicians in a range of cardiovascular, vascular, non-vascular, interventional and surgical procedures. The compact, cable-free C-arm system uses AI-powered guidance tools to help clinicians work efficiently and adapt to the spatial constraints of traditional interventional suites.
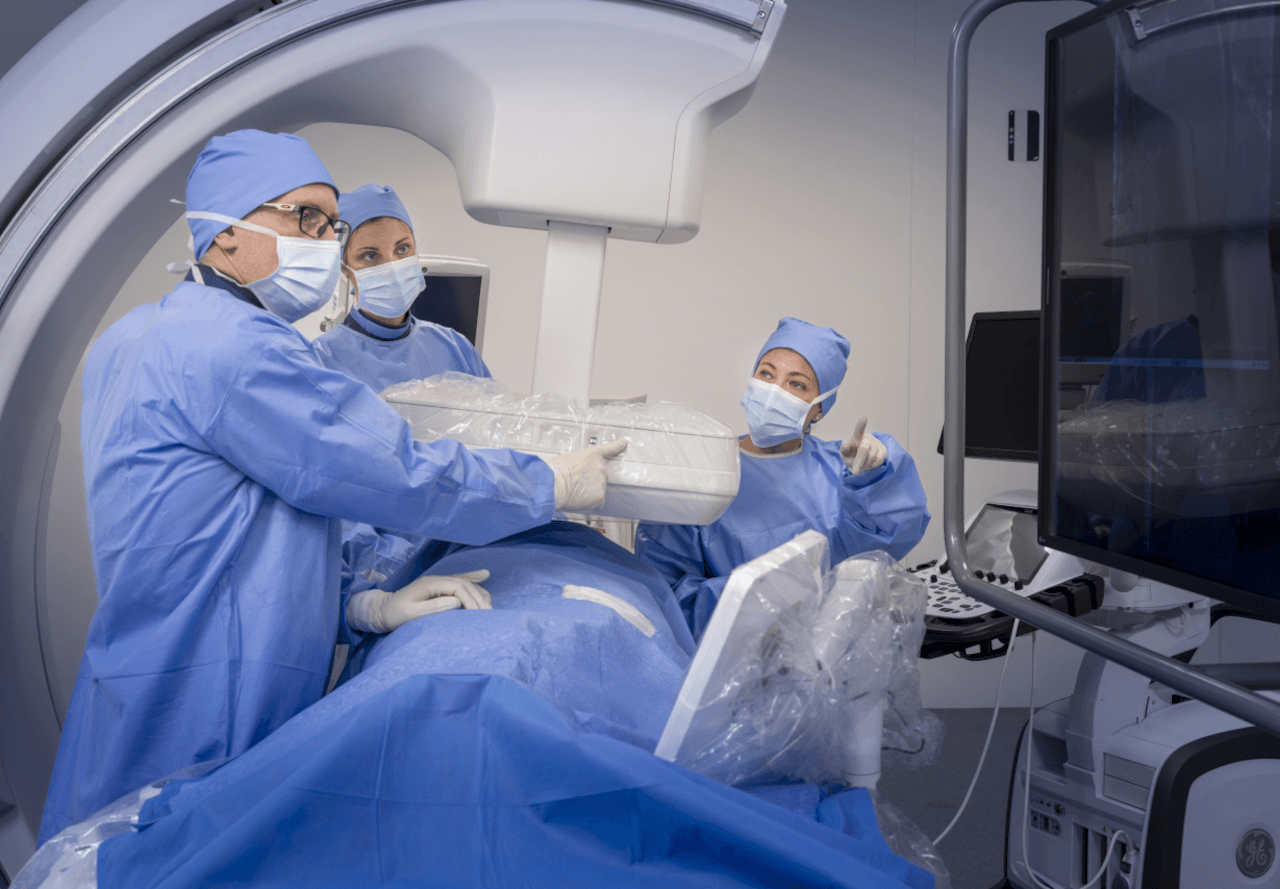
The Hôpital Marie-Lannelongue just South of Paris, France is a centre of excellence in thoracic, vascular and heart surgery, and national reference for complex aortic pathologies. It was the first medical centre in the world to have an Allia Moveo installed. The platform is intended to enable flexible cone beam CT and support the hospital in treating vascular and cardiology diseases. GE HealthCare says the manoeuvrable system is tailored to accommodate more patients regardless of body size or weight with a wide-bore design and effortless table panning to help care teams maintain comfort and access throughout procedures.
“With Allia Moveo, we had this opportunity to position the system very quickly in any working position that will adapt the best to us,” said vascular surgeon and the hospital’s head of aortic and vascular surgery Dr Stephan Haulon. “One of the major milestones of this new system is having access to the reconstruction of the cone beam CT with CleaRecon DL, which is AI driven and gives you much better image quality.”
CleaRecon removes streak artefacts caused by the pulsatile nature of blood flow in the arteries. The platform is also equipped with Motion Freeze, a tool to help offset involuntary respiratory motion artefacts, and metallic artefact reduction, designed to reveal anatomic details obscured by metal artefacts. Augmented guidance solutions include the Assist advanced clinical applications portfolio offering augmented guidance for complex procedures along with compatibility with other GE HealthCare and third-party systems.
Following the first global installation, Houston’s Baylor St Luke’s Medical Center saw the first installation in the USA.
Picture: the Allia Moveo system from GE HealthCare has received US FDA 510(k) clearance.
Published on page 4 of the March 2026 issue of RAD Magazine.